All research areas
No results


...about our contributions to the projects of other research groups
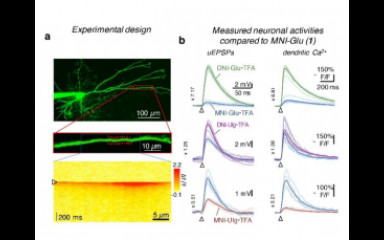
High efficiency two-photon uncaging coupled by the correction of spontaneous hydrolysis
In collaboration with the Szent Borbála Hospital in Tatabánya, we investigate brain samples of patients diagnosed with schizophrenia post mortem, which we compare with control subjects without neurological disease. The samples are fixed shortly after death and are therefore suitable for the detection of subtle structural abnormalities.

Cortex- wide activation of VIP- expressing inhibitory neurons by reward and punishment
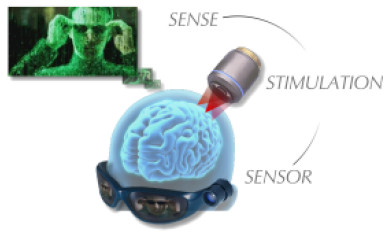
Restoration of visual perception by 3D artifical photostimulation
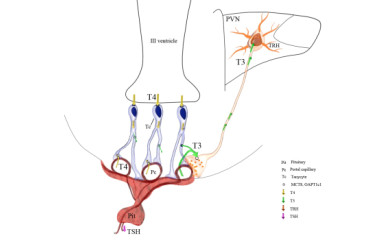
The hypothalamic-pituitary-thyroid (HPT) axis primarily functions to maintain normal, circulating levels of thyroid hormones that are essential for the biologic function of all tissues including brain development, regulation of cardiovascular, bone and liver function, food intake and energy expenditure among many others.

Most people experience at least one traumatic event during their lifetime. While the majority of individuals recover without long-term consequences, 10-30% of trauma-exposed individuals develop posttraumatic stress disorder (PTSD), a complex, severe and lasting mental condition which fundamentally decreases life quality. Therapeutic interventions in PTSD are insufficiently resolved to this day. Identification of individuals vulnerable to trauma-induced development of PTSD and treatment strategies selectively targeting this subpopulation represents a major clinical challenge with high therapeutic potential. Employing a translational laboratory animal model of trauma-induced lasting adverse behavioral changes, our group aims to identify behavioral markers of vulnerability and their neural correlates.
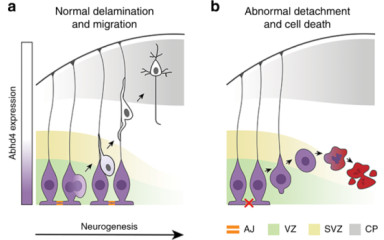
One of the major endocannabinoid molecules is anandamide. In this research project we aim to characterize its non-canonical synthesis enzymes, Abhd4 and Gde1, and to describe their diverse physiological functions in the brain.
With support of the European Framework Programmes (5-7), we have been collaborating with outstanding European research laboratories of this scientific field to elucidate novel neuronal and hormonal mechanisms that control metabolism centrally. These studies have explored several, new regulatory channels:
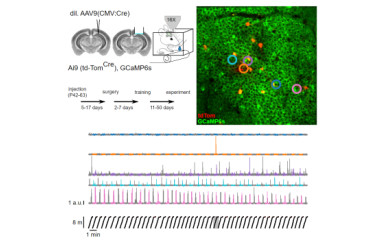
In vivo two-photon [Ca2+] imaging are performed in head-restrained mice while performing navigation in virtual reality to functionally characterize distinct pyramidal cells. This is followed by post hoc in vitro electrophysiological and anatomical experiments to reveal differences in intrinsic properties and synaptic innervation of the functionally characterized nerve cells.

Schizophrenia is a complex neurodevelopmental disorder that affects approximately 0.5%–1% of the global population and is 1 of the top 10 global causes of disability. Like most mental disorders, schizophrenia appears to be caused by a combination of genetic, environmental, and social risk factors linked to each other by epigenetic mechanisms regulating gene expression levels and molecular pathways. . Although the pathophysiology of schizophrenia is still not completely understood, there is a growing awareness that dysregulation of immune system components is fundamentally linked to the disorder.
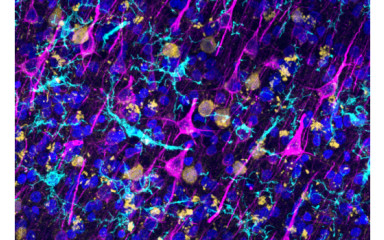
Central and systemic inflammatory mechanisms and the role of microglia in common neurological diseases.
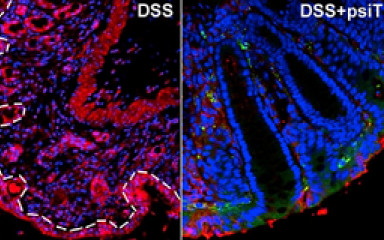
The aim of our research is to produce modified bacteria that can interfere with the synthesis of certain inflammatory cytokines or other inflammatory mediators in the mammalian host through trans-kingdom RNA interference. The model is that the modified bacteria encode specially designed small hairpin RNAs that are able to reduce inflammatory cytokine expression through RNA interference.
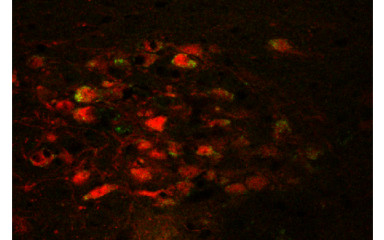
The neuroendocrine stress response is regulated by the hypothalamo-pituitary-adrenal system. The corticotropin-releasing hormone (CRH)-secreting neurons are located in the paraventricular nucleus of the hypothalamus which integrate stress information from other areas of the central nervous system and are able to trigger the appropriate hormonal stress response. However, during stress, autonomic functions, metabolism, immune processes and behaviour are also altered. The aim of our research is to elucidate the regulatory mechanisms and neuronal networks that coordinate elements of the stress response and to reveal the role of neurons expressing CRH in stress integration outside of the hypothalamus.
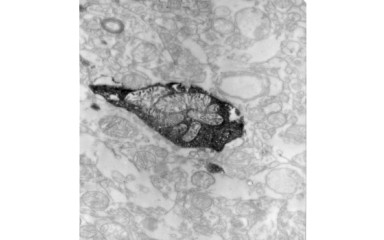
In this project, we study the synaptic organization of the human thalamus.
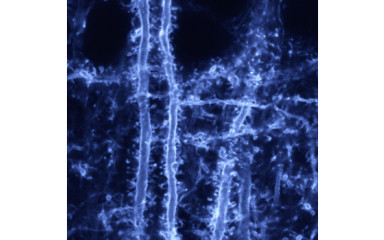
In this project we study the diversity of interactions between cortex and thalamus.
Active research and clinical experience have shown that the uncontrolled immune response to Sars-Cov-2 is characterized in many cases by a devastating cytokine storm (Chaolin Huang 2020). Morbidity and mortality are in part a consequence of this inflammatory response in the body (Ye Q 2020). The course of the disease is known to be more severe in pregnant women (Kathryn M Moore 2021), however, the effect of SARS-CoV-2 on the developing fetus is currently unclear. Case studies show that vertical transmission is rare, however, several studies support that placental and fetal infections may occur (Chen H 2020), (Dong L 2020), (Patanè L 2020). Several clinical manifestations of Covid-19 infection in the nervous system have also been reported recently (Desforges M. 2020), (Bohmwald K. 2018).

Determining the location and density of various voltage- and ligand-gated ion channels in defined subcellular compartments of hippocampal pyramidal cells, using quantitative LM and EM immunolocalization. Perform multi-compartmental modeling to generate functionally testable predictions of the functional consequences of specialized ion channel distributions. In vitro electrophysiology and imaging approaches are used to test the functional predictions of our models.
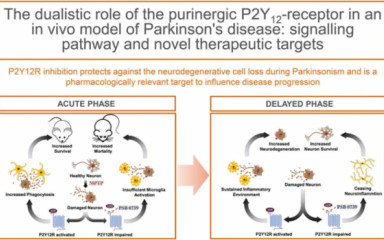
Parkinson’s disease (PD) is a chronic, progressive neurodegenerative condition; characterized with the degeneration of the nigrostriatal dopaminergic pathway and neuroinflammation.

Coping with different stressors is essential for survival and success, which is regulated by complex neural networks integrating cognitive and emotional processes. Under pathological conditions, excessive passive coping becomes detrimental, observed in anxiety and depressive disorders. Animal models can help to identify the neural substrates of individual vulnerability to develop such maladaptive conditions. We use rodent models (transgenic mice, early-life stress, cell type-specific manipulations) to explore the molecular, cellular, neuronal network level changes underlying anxious-depressive phenotypes.
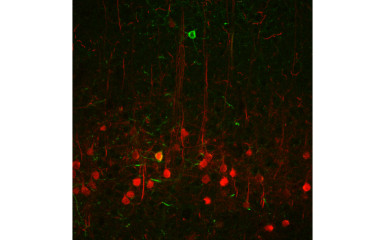
A novel research direction of our team is to unravel the circuit mechanisms underlying defensive behavior.
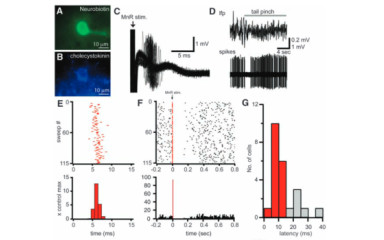
In vitro measurement of spontaneous neuronal network activity
In 2009 we described a novel form of modulation in the median raphe – hippocampus connection capable of selectively and rapidly recruiting a subset of inhibitory neurons. The function of this highly efficient form of modulation in shaping hippocampal representations is still unknown. In this project, we aim to unravel how the emergence and reorganization of hippocampal coding patterns linked to salient event is influenced by raphe-hippocampal rapid modulation.
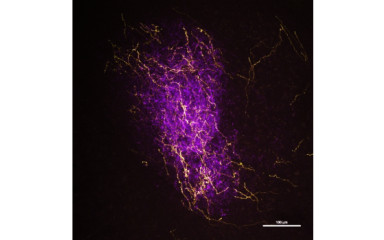
In this project we investigate the local connections within the basolateral amygdala.

It is known that thyroid hormone (TH) signalling is compartmentalized in the brain; glial cells are responsible for type 2 deiodinase (D2)-mediated TH activation, while neurons regulate their intracellular TH levels by type 3 deiodinase (D3) catalyzed inactivation. Little is known however on how TH are presented to neurons and how the active and inactive forms of TH are transported between cells and brain regions.
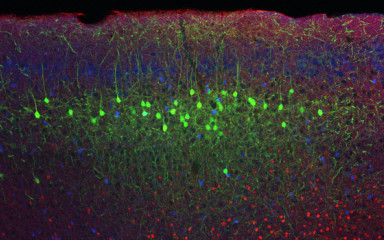
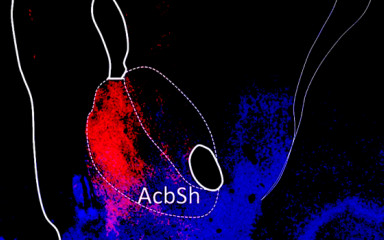
Investigating neural circuits that control reward learning is crucial if we want to understand healthy behavior or disorders like addiction.

Disruptions in a child's social environment, such as instances of abuse or neglect, can significantly modify the development of the brain. This alteration serves as a potential risk factor for the emergence of later psychopathological disorders, particularly those related to social behavior within psychiatric conditions. Using different translational, clinically relevant models of early social stress (abuse, neglect), we aim to reveal alterations in maturation processes of regions implicated in emotional control and their long-term functional (behavioural) consequences, allowing the understanding of cellular mechanisms and identification of novel molecular targets.
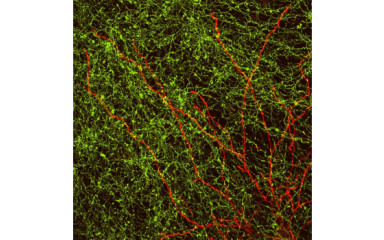
In this project we study the role of thalamus in motor learning.

During development, the negative thyroid hormone (TH) feedback setting ("set-point") of the hypothalamo-pituitary-thyroid (HPT) axis is fixed for the entire life-span, but TH levels required for optimal tissue function vary in an age-dependent manner. However, the feedback regulation of the HPT axis is not able to adapt to this changing demand by changing the fixed set-point, that may contribute to age-dependent impairments of tissue function. Our aim is to investigate whether there are mechanisms that may contribute to age-dependent regulation of tissue-specific TH homeostasis despite the fixed set-point of the HPT axis.
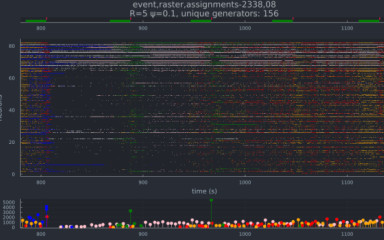
Coding by transiently emerging co-active ensembles of neurons has long been a central tenet of neuroscience formulated in the influential assembly hypothesis. Accordingly, the animal’s actions can be predicted much more efficiently from the coordinated activity pattern of neurons than from the stimulus or event-locked spiking of single units. In contrast to cortical and hippocampal networks, assembly coding in subcortical modulatory circuits is almost fully unexplored. In this project, we aim to reveal and characterize the assembly code in the median raphe (MR), the source of ascending serotonergic neuromodulation of the limbic system. We expect to identify a fundamentally novel mode of neuromodulation whereby adaptive behavioral responses are controlled by the correlated activity of modulatory neuronal assemblies.
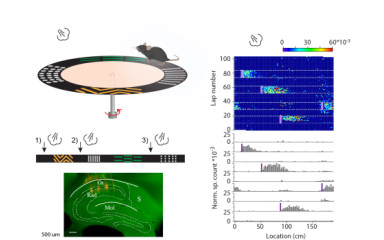
Throughout life animals inevitably encounter unforeseen threatening events. Activity of principal cells in the hippocampus is tuned for locations and for salient stimuli in the animals’ environment thus, forming a map known to be pivotal for guiding behavior. Here, we investigate if a code corresponding to threatening stimuli exists in the CA1 region of the dorsal hippocampus.

Hypoxic-ischaemic encephalopathy (HIE), a condition caused by oxygen deprivation around birth, accounts for the death of around one million newborns worldwide every year, and the number of neonates who suffer permanent neurological damage is much higher. Even with a mild course, HIE can lead to long-term neurodevelopmental, behavioural, cognitive and psychiatric disorders. The aim of our research is to identify potential new therapeutic targets and biomarkers that may predict increased risk for subsequent psychiatric disorders by translational preclinical investigation of the brain disease mechanisms underlying the long-term effects of HIE.

With our research activity we aim to contribute to the understanding of how distinct endocannabinoid-related signaling pathways regulate certain forms of synaptic plasticity.
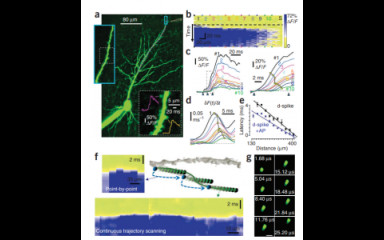
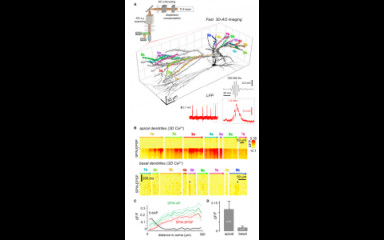
Fast, two- photon imaging with three-dimensional scanning of spine, dendritic and neuronal assemblies in behaving animals
Prefrontal cortex plays a role in many higher order cognitive processes.

Depression - diagnosed as a condition lasting at least 2 weeks, with lethargy and/or loss of pleasure as the leading symptoms - is the most common psychiatric disorder in the world.
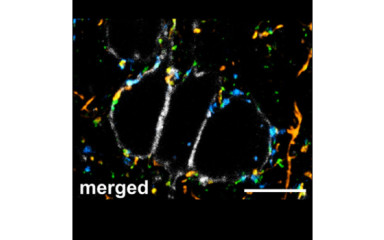
…about the roles of small axons, whose size matches that of the majority cortical axons
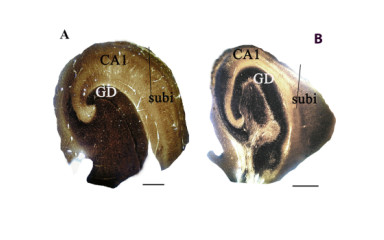
Our laboratory investigates cortical tissue removed due to therapy-resistant epilepsy and compares it with a short post-mortem interval sample from control subjects without neurological disease. The process is described in the "Methods" tab.
The three main causes of therapy-resistant epilepsy are tumour, hippocampal sclerosis and focal cortical dysplasia, and most commonly develop in the temporal lobe. Our group and its predecessor described several alterations of interneurons and their connections in temporal lobe epilepsy with or without hippocampal sclerosis. Currently, our main area of investigation is epilepsy associated with focal cortical dysplasia. In this context, we have described alterations in parvalbumin-containing perisomatic innervation and we intensively investigate impairments of the cannabinoid system, microglia and blood-brain barrier elements in the disease.
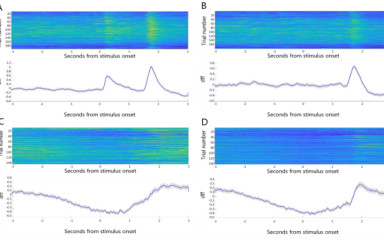
In vivo examination of age-dependent changes in the activity of basal forebrain cholinergic neurons during a Pavlovian conditioning task
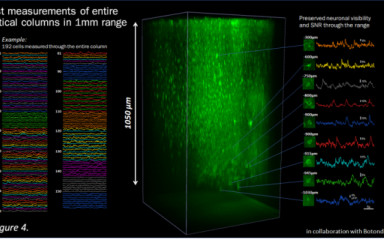
Fast 3D imaging in behaving mice
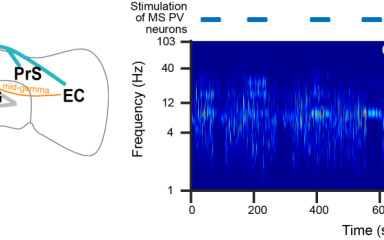
The medial septum modulates hippocampal oscillations beyond the theta rhythm
Gonadotropin-releasing hormone (GnRH)-synthesizing neurons represent the final output pathway of the hypothalamus in the neuroendocrine control of reproduction. Pulsatile GnRH secretion into the hypophysial portal circulation regulates the synthesis and release of the two pituitary gonadotropins, LH and FSH, which in turn, govern gonadal functions. Gonadal sex steroid hormones exert positive and negative feedback effects on the neurosecretory output of GnRH neurons via mechanisms that are poorly understood. A major research focus of the Laboratory of Endocrine Neurobiology has been on the neuronal and hormonal mechanisms that regulate GnRH neuronal functions.
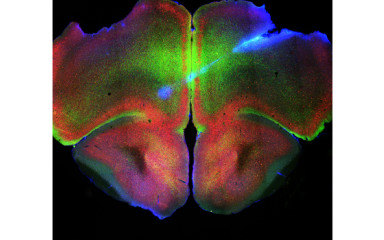
The midline thalamus has a key role in the regulation of working memory, cognitive processing and sleep-wake transition, as well as in controlling cortical oscillations.
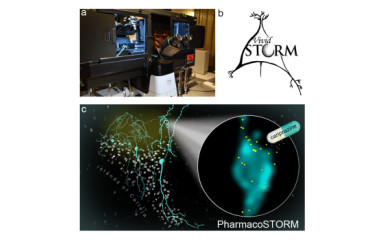
Our latest paper in Nature Communications presents the PharmacoSTORM method developed in our laboratory.

ASD is a complex neurodevelopmental condition caused by interactions of environmental and genetic factors. Recently, maternal viral infections during pregnancy and their immunological consequences have received particular interest in research as risk factors for neurodevelopmental disorders.
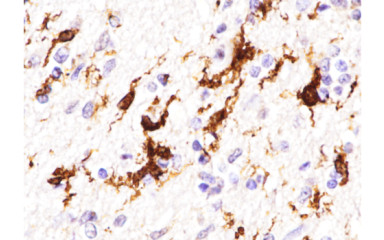
The neurological and neuroimmunological aspects of SARS-CoV2 virus infections.
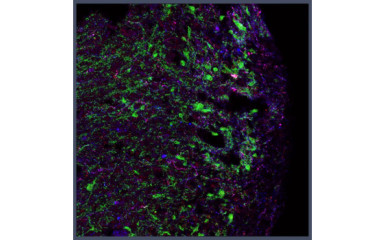
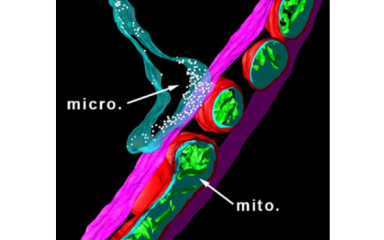
Investigation of the molecular anatomy, function and mediators of microglia-neuron and microglia-vascular interactions.
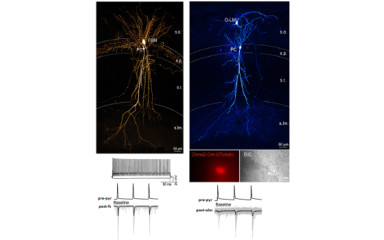
Determine the molecular specializations underlying the functional diversity of synapses, such as the probability and short-term plasticity of neurotransmitter release. In vitro electrophysiology, two-photon imaging, LM and EM immunolocalization are combined to address these issues.
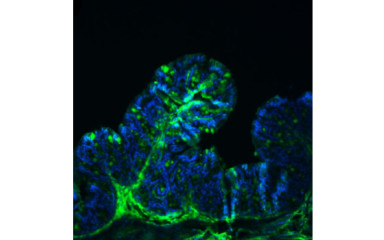
Interface between the body and the environment are populated by a large number of microorganisms, which are called the microbiome. The substances produced by the microbiome, their specific metabolites, have a significant influence on the host. The largest and most complex community of microorganisms in the body is the gut microbiome, which is made up of approximately 1015 microorganisms and is estimated to contain at least 1500-2000 different species. The composition of the microbiome is influenced by many factors, the way of birth, diet, antibiotic treatment, etc. Intake of live bacteria (probiotics), intake of specific bacterial nutrients (prebiotics) and their combination (symbiotics) may also influence of the gut microbiome.
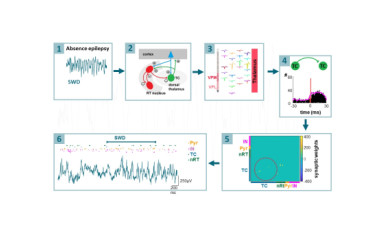
We would like to identify those elements of the thalamocortical circuitry which are involved in the initiation and maintenance of epileptic events

Environmental pollution is rapidly increasing the amount of substances known as endocrine disruptors, that potentially exert a disruptive effects on hormone homeostasis, including thyroid hormone (TH) economy. These among others include plastic additives, pesticides, veterinary products, and their effects can vary widely.
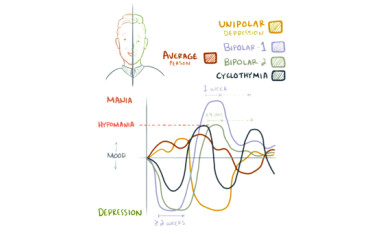
Purinergic dysfunction are involved in the pathological process of mania and depression, but the essential association is not fully understood. The activation of the purinergic P2X7 receptor (P2X7R) plays a central role in inflammation, microglia activation and IL-1β release and activates NLRP3 inflammasome. We investigated whether the d-amphetamine (AMPH) has an inflammatory profile, and the P2X7R has an effect via IL-1β in this model.
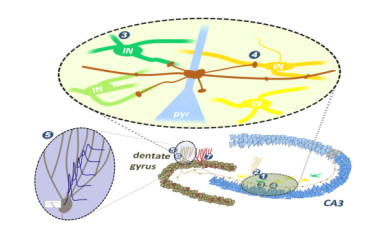
…about the cellular interface between DG and CA3 regions
Subcortical modulation is an indispensable component of cortical function, and ultimately, is key for adaptive behavioral responses. Therefore, disruption of subcortical modulation leads to debilitating psychiatric conditions. A key but largely ignored area of research concerns the control of subcortical modulation by cortical feedback. In this project we explore how the top-down control of the median raphe by the prefrontal cortex controls memory-guided behaviors. Our hope is to identify a missing link in the process that leads from normal subcortical function to pathological cortical operation.
While the HPT axis warrants the stability of circulating thyroid hormone levels, a complex machinery of thyroid hormone transporters, metabolizing enzymes, receptors and co-regulators ensures that the local thyroid hormone action meets the highly variable requirements of the different tissues.

Our aim is to identify the basic cellular and molecular pathways that underlie the symptoms of hypothyroidism in a group of patients who respond poorly to thyroxine (T4) monotherapy. While the therapy is able to normalize the TSH of these patients, they still suffer from symptoms of tissue hypothyroidism that adversely affect cognitive function and regulation of energy homeostasis.
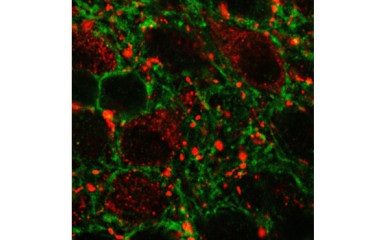
Glucagon-like peptide-1 (GLP-1) is an incretin hormone. It is derived from the posttranslational processing of proglucagon. This prohormone is synthesized by three cell populations, the neuroendocrine L cells of the intestinal mucosa, the ß cells of the pancreatic Langerhans islands and in a neuronal population located in the nucleus tractus solitarii (NTS) and intermediate reticular nucleus of the medulla oblongata.
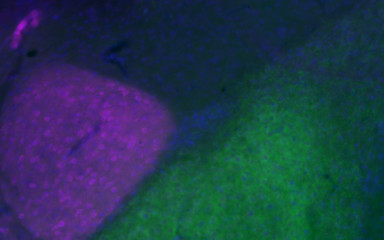
The amygdala is a central element of the brain’s emotional network, but there are a lot of unresolved questions around this brain region.

The prevalence of anxiety disorders is significantly the highest among all mental illnesses, estimated at around 34%. Despite its high prevalence and serious individual and societal impacts, the neurobiological mechanisms underlying this group of disorders are not fully understood. Consequently, the currently available pharmacotherapeutic treatments are ineffective in 40% of cases. Furthermore, promising new drug targets identified in preclinical studies prove ineffective in human clinical trials at an alarming rate. Our hypothesis suggests that preclinical anxiety tests can only measure transient anxious states that are highly influenced by environmental impacts, rendering them unsuitable for modelling high trait anxiety, a core symptom of anxiety disorders.
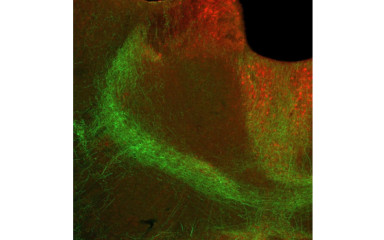
In this project we study the role of thalamus in stress induced behavioral alterations.
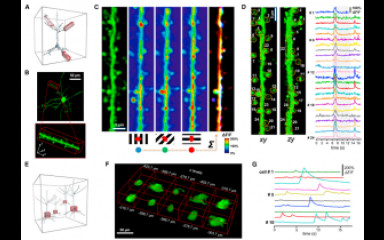
The sex hormone 17β-estradiol (E2) is primarily synthesized in maturing ovarian follicles. Cyclic changes in serum E2 levels across the menstrual cycle exert profound effects on reproductive tissues in women. E2 also plays an important role in the maintenance of normal limbic and cortical functions. Around menopause, when E2 levels decline the incidence of cognitive and mood disorders increases, which can be prevented with hormone replacement therapy. A major research interest of the Laboratory has been in the molecular mechanisms whereby E2 preserves good mood, capability of learning and processing memory via interactions with cortical and limbic structures. The classic actions of E2 are mediated by two estrogen receptor isoforms, ERα and ERβ. They are ligand-dependent transcription factors which regulate gene expression in the presence of E2. Prefrontal cortex (PFC) and the hippocampus are known targets of steroid hormone signaling.


